Other technologies that have shaped the market structure have been the presence of the technologies like the SMARt-Ig and ARt-Ig. The marketing of the monoclonal antibodies is also leading to high market demand as it brings in fresh investment and allows manufacturers to merge and alliance. Tychan another startup is using convergence analytics to fasten the biologics process for the human trial. Startups are rapidly developing technology platforms and space to handle the COVID-19 situation better. Yurogen Biosystems–USA-based startup is manufacturing humanized mAbs. Pluvia, FerroSens, and Adcendo are a few of the startups developing therapeutics antibodies. Nippon Kayaku and Mitsubishi Gas Chemical in 2016 announced to start the production of mAb drugs in Niigata.Įurope’s leading startups are also revolutionizing the antibody production domain to develop novel therapies and treatments. The expansion of the market is also reliable on the new manufacturing facilities dedicated to antibody production. Companies like Takeda Pharmaceutical are developing plasma-derived antibodies for COVID-19 treatment taking a different route from the laboratory-made antibody. The manufacturers of the antibodies are participating in the rigorous launch of the antibodies to treat diseases like plaque psoriasis. WuXi Apptec in the year 2012 inaugurated the monoclonal antibody seminar on a global scale. The market’s growth is also highly influenced by the summits presented by the manufactures investing in the biologics and production capacities to expand the biologics domain. Harbour Biomed in July 2020 raised over US$300 million to support the advancement of the preclinical molecules. Alexion, Forty-Seven, and Harbour BioMed are a few of the startups in this space. Thus higher investment in these startups and companies can also lead to the development of advanced therapies to cater to cancer patients. The antibody-based immunotherapy startups are also being glorified as equipping immunotherapy with antibody therapy which can deliver substantial results for successful cancer treatment. 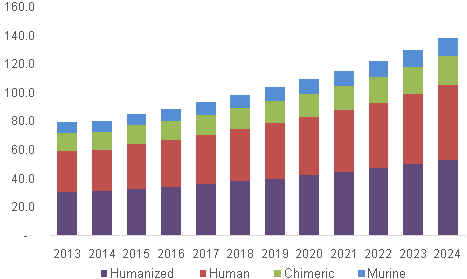
Firms such as Mabwell Biotech raised RMB 1.97 Million to expand their production of biopharma products. The market is also benefitted from the investment secured by the startups operating in the biopharmaceutical space. In 2012, the mAb drugs were the top demanded drugs in the USA, and recorded revenue in billion dollars. The growing investment in monoclonal antibodies will create a huge market investment pool for the anti production market in general. The monoclonal antibodies are classified as the main driver of the biotech industry and also lead the market for therapeutic drugs. Software tools like the PEAKS AB have been deployed to understand the characteristics of the monoclonal antibodies. 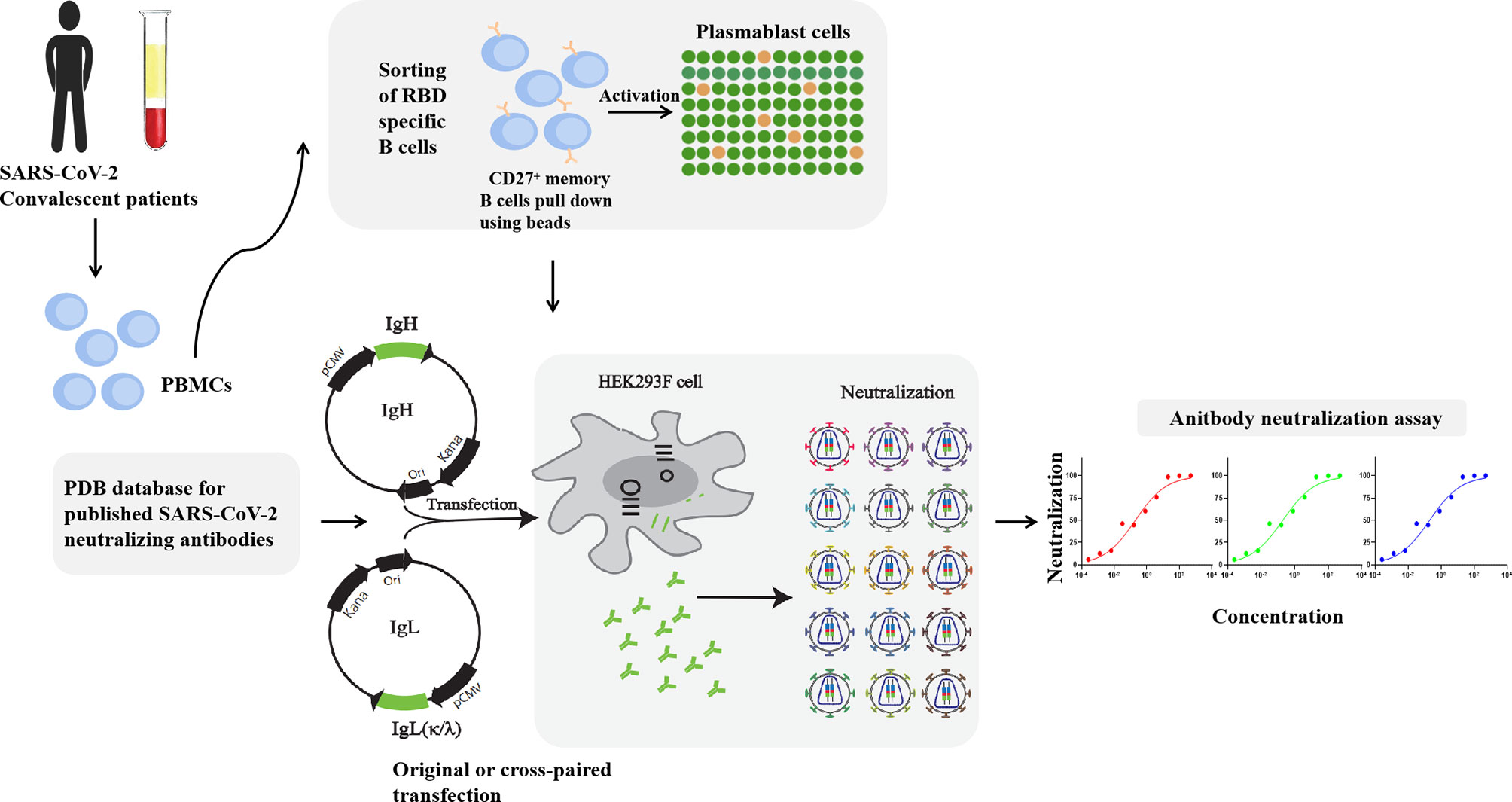
The therapeutic indications included asthma, multiple sclerosis, and hemorrhage among others.Īs the market moves in the direction of advanced technology, software tools are becoming important in solving the complex structure of the antibodies. In 2017 also, various monoclonal antibodies were cleared and approved for therapeutic use by the FDA and EMA. In November 2020, the FDA approved and authorized the use of mAb therapy to reduce the burden on the healthcare system. The use of the mAb therapy has also been rendered effective in the treatment of COVID-19 patients and such therapy has also received FDA approvals to better serve public health. These antibodies equip the body’s immune system to combat cancer. The rise in the lifestyle concerning diseases like cancer also has the potential use of monoclonal antibodies (mAb). Clinical research’s importance in developing diagnosis and medical devices is rapidly growing as the world faces an increasing burden of lifestyle concerning diseases and the spread of the novel COVID-19. 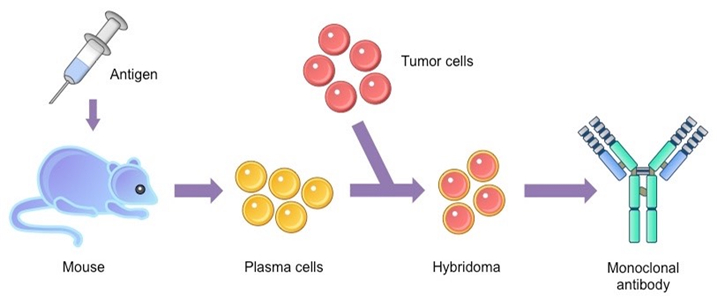
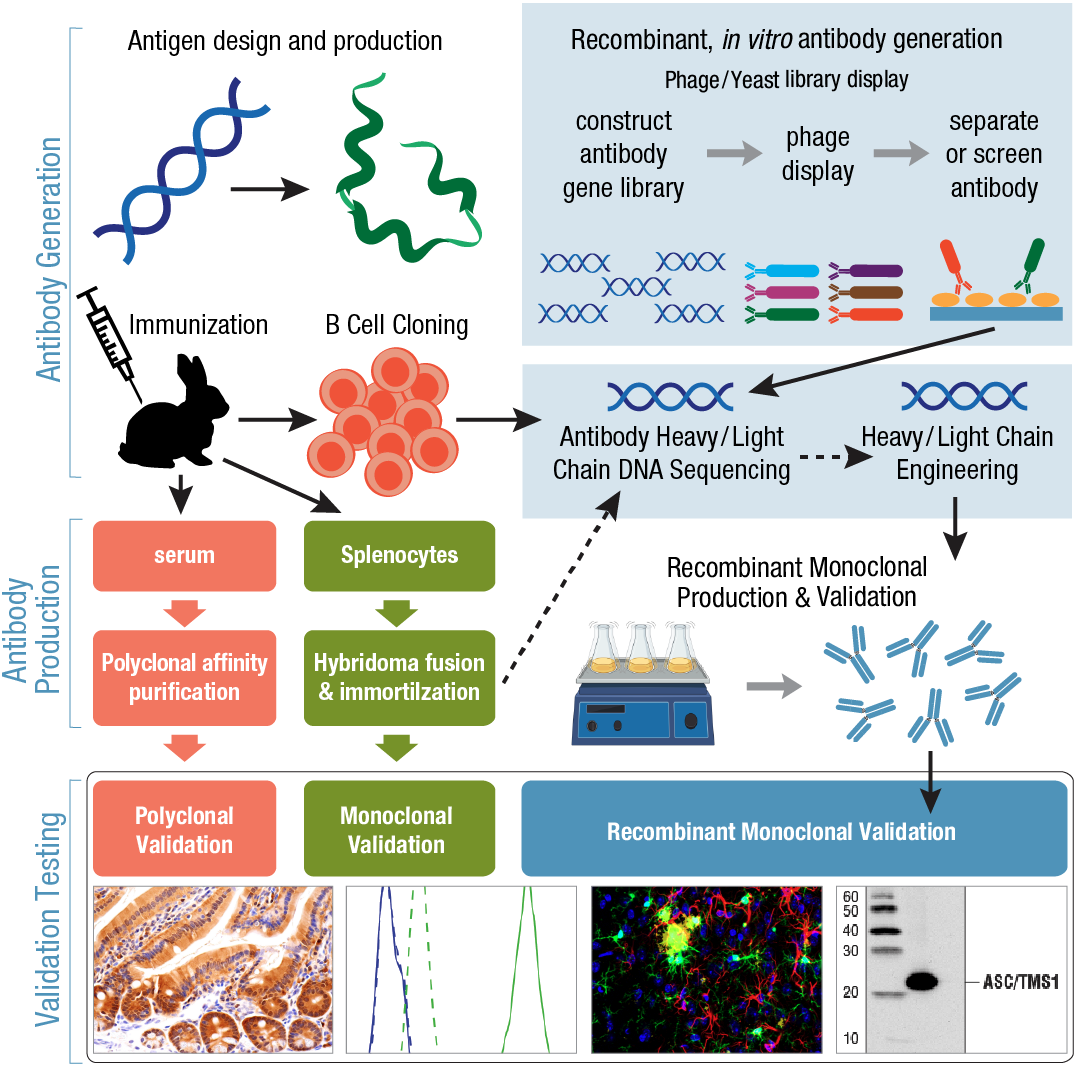
The production of the antibodies is critical from clinical medicine and biological research point of view. The use of hybridoma technology helps in the production of monoclonal antibodies. The development of hybridoma technology for antibody production is widely used. The antibody production domain has experienced technological expansion since 1975. The process of antibody production involves the process of developing certain antibodies that require the use of immunization, hybridoma production, immunogen preparation, purification, and screening.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
